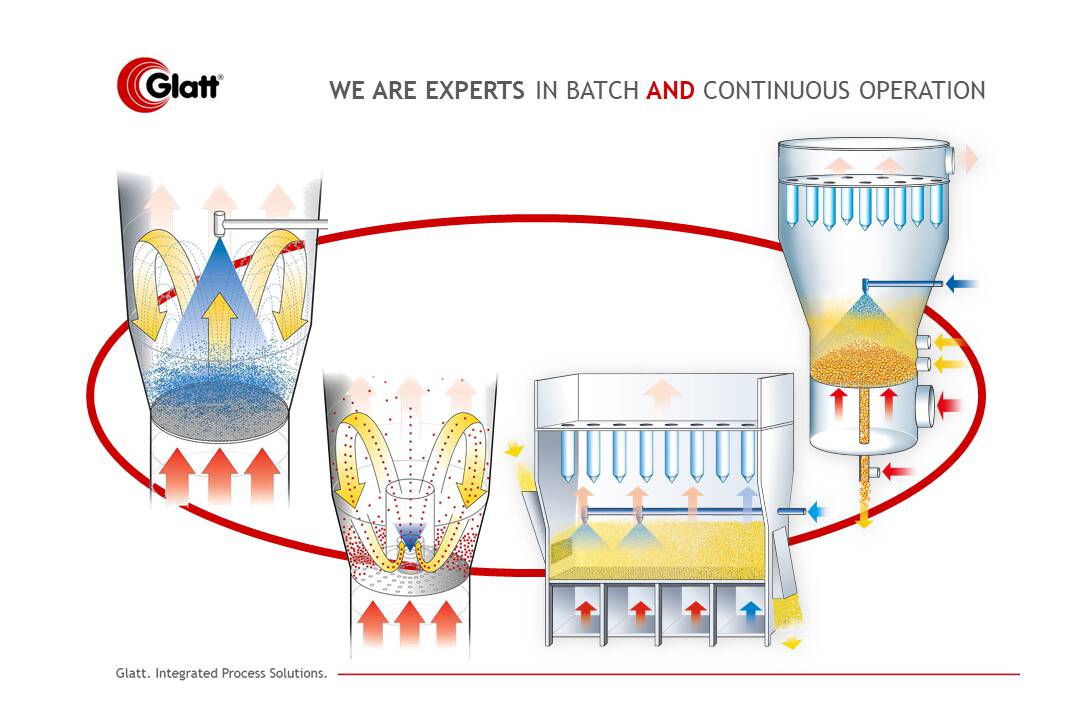
Plant-based functional ingredients behave differently than manufacturers of nutraceuticals and cosmetic products would like. Innovative processes such as double microencapsulation can help them to overcome bioavailability and solubility challenges.
Among the currently trending functional ingredients, interest in natural sources such as turmeric and its polyphenol, curcumin, has been increasing for years. The spice is not only in demand in the culinary world, but has also found great promise in both consumer and scientific applications.*1 Hewlings and Kalman reported that the reason for this is because of the medicinal properties of this rhizomatous perennial plant (Curcuma longa) from the ginger family, which have been known for thousands of years.*2 In Asian countries, turmeric has traditionally been used as a medicinal plant for its antioxidant, anti-inflammatory, antimutagenic, antimicrobial and anticancer properties.*3–*5 This sought-after ingredient is not only used in foods, beverages and nutraceuticals, but also in cosmetic products. One of its main formulation challenges, however, is its poor bioavailability, which is thought to result from limited absorption, rapid metabolism and excretion.*6 One way to improve the bioavailability of nutrients is to add certain agents; in the case of curcumin, for example, piperine is recommended. However, this pepper-derived ingredient can also cause discomfort.
A more promising solution is a technological approach that specifically optimises the properties of the required substance for the purpose of use. Glatt Ingenieurtechnik, an expert in plant engineering and process development, specialises in issues such as these. Known as a pioneer of fluidised bed technology, Glatt offers, among other things, feasibility studies on flexible plant concepts that enable various particle formation and functionalisation processes in both continuous and batch mode. Together with the expertise of Sphera Encapsulation — who provided proprietary formulation know-how (SpherAQ) — and Lipofoods, who contributed to the development of this new encapsulation approach, a challenging project was tackled.
The double encapsulation approach
As mentioned above, the development of curcumin supplements and medicinal preparations is limited because of the low solubility of the compound in water and the associated poor absorption in the intestine when administered orally.6 Moreover, curcumin is also known for its intense colouring power and its peculiar taste (which is not always appreciated by consumers). Furthermore, the raw material cannot be dissolved in aqueous liquids, which limits the ability to use it in most of foods and beverages (Figure 1).
The new process, a double encapsulation approach, first adds functionality to individual curcumin particles; then, in a second step, it enhances particle formation. During the first step, microparticles are produced using an innovative system that combines the advantages of a biodegradable and highly nutritious, protein-based hull with a defined (small) particles size that guarantees transparency, high stability in solution and excellent bioavailability. Neither aggregation nor ...
Overcoming solubility issues
Many processes can be used to dry liquids. As such, it makes sense to evaluate the desired properties of the final product and whether a powder or a granulate is preferred. This then prompts the question: what influence does the process have on product quality and its application properties? Liquids with a medium to low solids concentration are normally dried in highly efficient spray towers, which produce large volumes of powder using a high-temperature processing gas and very short residence times. However, if specific product characteristics such as bulk densities, good dosing properties or improved solubility are required, further process steps are needed. Here, the normally effective spray drying process quickly becomes a complex multistage procedure.
At this point, spray granulation — especially spouted bed technology — offers several benefits. (Figure 2). By continuously atomising the liquid, whether it’s a solution, dispersion or emulsion, particles form and grow in a fluidised bed to the required size. Shape, density and other properties can be adjusted by varying the operational parameters. In this project, spouted bed technology provided the notable advantages of shorter residence times and, thus, smaller particles within very narrow tolerances. Furthermore ...
You can access the complete article via the adjacent link.
Co-operative process development
Nutraceutical Ingredients Lubrizol Life Science (formerly Lipofoods) created Curcushine microcapsules in collaboration with Sphera Encapsulation. The technology applied to the molecule is called SpherAQ, which enables the creation of high-quality encapsulates that are totally soluble in water and can be consumed without the addition of any flavours or aromas.
The process was initially developed at laboratory scale, whereby one challenge was to co-ordinate the individual process steps into a sequence that was suitable in terms of time and throughput. For this purpose, the stability of the intermediate products and the effect of changing the spray solution parameters on the final product had to be evaluated.
This configuration was tested several times at pilot scale before a transfer to production-size equipment could be considered. Here, too, the various process steps had to be adapted to work with larger system components to ensure that the continuous drying process was both safe and efficient.
Besides the physical properties of the product, the curcumin extract also benefited from ...
References
- J. Hewlings and D.S. Kalman, “Curcumin: A Review of Its Effects on Human Health,” Foods 6(10), 92 (2017): doi: 10.3390/foods6100092.
- I. Priyadarsini, “The Chemistry of Curcumin: From Extraction to Therapeutic Agent,” Molecules 19(12), 20091–20112 (2014): doi: 10.3390/molecules191220091.
- L.A.D. Lestari and G. Indrayanto, “Curcumin” in H. Brittain, Ed., Profiles of Drug Substances, Excipients and Related Methodology, Volume 39 (Elsevier, Cambridge, MA, US, 2014) pp 113–204.
- C. Reddy, et al., “Curcumin for Malaria Therapy,” Biochemical and Biophysical Research Communications 326(2), 472–474 (2005).
- E. Wright, et al., “Bioactivity of Turmeric-Derived Curcuminoids and Related Metabolites in Breast Cancer,” Current Pharmaceutical Design 19(34), 6218–6225 (2013).
- Anand, et al., “Bioavailability of Curcumin: Problems and Promises,” Molecular Pharmaceutics 4(6), 807–818 (2007).
Authors
- Gudrun Ding, Head of Business Development, Process Technology, Glatt Ingenieurtechnik
- Francesca Zanoni, Research and Development Manager, Sphera Encapsulation,
- Isabel Gómez, Global Marketing Manager, Nutraceutical Ingredients Lubrizol Life Science, Health
originally published in the magazine ‘Nutraceutical Business Review’, issue August/2021, HPCi Media Limited